|
I decided it would be especially useful (and distributable) if I added a GUI, and this allowed me to learn tkinter at the same time. I figured it would be useful to have a quick application to access that would calculate it for me, especially one that would determine the limiting reagent for me. Add an exception handler, in the case that a chemical formula AND molar mass are entered for any reagent or the product, to determine if they are close enough (a button that allows you to add as many reagents as participated in the reaction, and let the application determine the limiting reagent from there. The calculator prioritizes the chemical formula for calculating molar mass (through the periodictable library). The application accepts a chemical formula or molar mass for the limiting reagent, as well as the mass used, as well as the chemical formula or molar mass and weight of the product recovered. No need to do manual calculations with yield formulas when you have this calculator. It gives you the easiest way to find the value of yield of a theoretical equation. Graphical application coded in python utilizing Tkinter GUI framework and 'periodictable' library. Percent yield calculator is a very useful tool for students of chemistry. If the theoretical yield is 2.0 g, calculate the percentage yield of copper sulfate. How to use our Theoretical Yield Calculator?Įnter the percentage yield and actual yield found by using the formulas written above in their respective sections and click calculate.GUI Percent Yield calculator for chemical experiments Learn about and revise chemical calculations with this BBC Bitesize GCSE Chemistry (Edexcel) study guide. Mole is equal to mass over the molar mass. The actual yield can easily be calculated by this formula In the laboratory, it is the amount of product that is formed in your beaker, after it is purified and dried. It is the amount of product obtained when the reaction is actually performed. All you have to know is the percentage yield and actual yield. If you use our Theoretical Yield Calculator, you can easily calculate the theoretical yield. Most of the time we are asked to find the theoretical yield from actual and percentage yield. It is not possible to convert all of the reactants completely into the required product without producing any by-products or preventing any side reaction to take place. No laboratory reaction is a perfect chemical reaction. It is the calculated product without performing any experiment. In the above-mentioned example, the excess reactant is patties. Less than 10 of by-products were left in this chemical reaction resulting in a low amount of waste making the method an efficient one. To determine the percent yield and identify how effective the chemical reaction is, multiply the decimal percentage of 0.91244 by 100. It is the reactant of which some quantity is left by the end of the reaction. The decimal percentage of percent yield is 0. The limiting reactant in this process is buns. You can make on only 23 sandwiches although you are left with one extra patty. 
For example, you have 47 buns and 24 patties. If you know which is reactant is limiting, you would use only the required amount of other reactants. In all the previous calculations we have performed involving balanced chemical. In a chemical reaction, the yield or output, i.e. It helps in increasing the yield of a reaction. Define and determine theoretical yields, actual yields, and percent yields. The calculation using the theoretical yield formula is simple. One can calculate the value of one variable by knowing the value of the other two variables. It is the reactant that decides the amount of product obtained. The formula used to calculate percent yield is as follows: Percent yield () (Actual yield / Theoretical yield) X 100 To calculate the percent yield, you must know the value of Actual yield and Theoretical yield. 
For example, Consider the decomposition process of calcium carbonate. 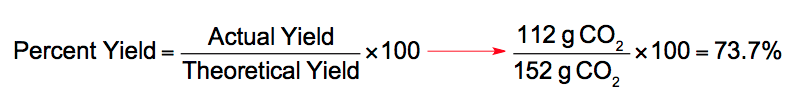
P (A / T) × 100 Where, P is the percentage yield, A is the actual yield, T is the theoretical yield. It is the product of a perfect chemical reaction. If the actual and theoretical yields are equal, the percent yield is 100. It is the amount of product obtained when the whole of the limiting reactant used in a reaction is completely converted into the products without any loss of reactants. This chemistry video tutorial explains how to calculate the percent yield, actual yield and theoretical yield of a product produced in a chemical reaction gi.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
